Investigating an Alternative Approach to Cancer Immunotherapy: Eliminating Immune System-suppressing Treg Cells
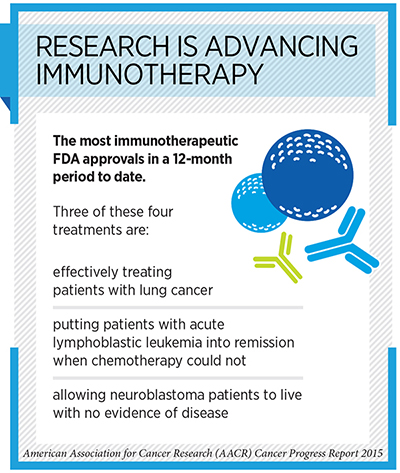
The number of anticancer immunotherapeutics approved by the U.S. Food and Drug Administration (FDA) is rising rapidly. In fact, the four anticancer immunotherapeutics approved by the FDA in the 12 months covered by the recently released AACR Cancer Progress Report 2015—Aug. 1, 2014, to July 31, 2015—was the greatest number of anticancer immunotherapeutics approved by the agency in any 12-month period to date.
Much of the media attention has focused on FDA-approved anticancer immunotherapeutics like pembrolizumab (Keytruda) and nivolumab (Opdivo), which are referred to as checkpoint inhibitors. As I have explained in a previous post on this blog, these agents work by releasing brakes on immune cells called T cells, which have the natural potential to recognize and eliminate cancer cells. Research has shown, however, that triggering brakes on T cells is just one way in which tumors can evade destruction by the immune system.
A study just published in the AACR journal Clinical Cancer Research reported results of a phase I clinical trial in which researchers investigated targeting another mechanism by which tumors are thought to avoid cancer-fighting T cells. Specifically, the researchers sought to eliminate cells—called Treg cells—that can inhibit anticancer immune responses.
What are Treg cells and why are they a potential target for cancer treatment?
Regulatory T cells, so-called Treg cells, are a subset of T cells that are defined by expression of two proteins, CD4 and FoxP3. These cells are critical for keeping other immune cells in check; they help prevent the immune system from attacking normal cells and tissues, causing autoimmune disorders.
Preclinical studies have shown that Tregs can also prevent the immune system from attacking tumors and clinical research has shown that these cells accumulate in the tumors of some patients with cancer. These observations led to the idea that eliminating Tregs in patients with cancer might unleash the natural potential of the patient’s cancer-fighting T cells, and to several clinical trials testing this hypothesis.
The Clinical Cancer Research study
In the phase Ia clinical trial reported in Clinical Cancer Research, the researchers evaluated whether using a therapeutic antibody called KW-0761 (mogamulizumab), which targets the protein CCR4, would eliminate Tregs in the blood of patients with lung or esophageal cancer. KW-0761 has been approved in Japan for the treatment of relapsed or refractory adult T-cell leukemia/lymphoma.
One of the senior authors on the paper, Ryuzo Ueda, MD, PhD, professor in the Department of Tumor Immunology at Aichi Medical College in Nagoya, Japan, explained in a news release that they used KW-0761 because activated FoxP3+CD4+ Tregs that accumulate in tumor tissue have been shown to express CCR4 molecules on their surface.
The research team enrolled seven patients with non–small cell lung cancer and three patients with esophageal cancer in the clinical trial. After analyzing blood samples obtained before the first treatment with KW-0761 and then every four weeks, they found that the number of FoxP3+CD4+ Tregs in the blood of all patients was dramatically reduced following treatment with KW-0761.
There were no dose-limiting toxicities and most adverse events were grade 1 or grade 2, with skin-related adverse events occurring most frequently.
“We were pleased to see that infusion of even a small amount of the KW-0761 efficiently depleted Tregs from the peripheral blood for a long time [several months],” said the co-senior author on the study, Eiichi Nakayama, MD, PhD, a professor at Kawasaki University of Medical Welfare in Kurashiki, Japan. “Unfortunately, we observed only a modest induction of antitumor immune responses and no marked clinical responses with KW-0761 monotherapy.”
What happens next?
Nakayama explained that the research team is planning to investigate whether combining Treg depletion with other immunotherapies, such as checkpoint inhibitors, can augment the antitumor immune response in patients with cancer.
However, the authors of a commentary published in Clinical Cancer Research in June 2015 emphasize that we still have much to learn about the role of Tregs in the initiation, development, and progression of different cancer types. They note that some studies point to these cells as not always being “bad guys,” and potentially even helping prevent cancer from developing in some cases by keeping in check inflammation, which can drive cancer initiation.
We hope that we will learn more about this exciting area of cancer immunology and immunotherapy at forthcoming meetings, like the AACR Annual Meeting 2016, which has an abstract submission deadline less than two months away: Tuesday, Dec. 1, 2015.





