
Is Chemo-free Treatment Coming for HER2-positive Breast Cancer?
Evidence grows that some people with HER2-positive breast cancer may do well on HER2-targeted therapy alone.

Evidence grows that some people with HER2-positive breast cancer may do well on HER2-targeted therapy alone.
Physicians consider which patients are ideal for immunotherapy drug combinations.

To learn more about cancer prevention and risk reduction, we talked to Raymond N. DuBois, MD, PhD, FAACR, co-editor...
The CAR T-cell therapy lisocabtagene maraleucel was approved for the second-line treatment of certain patients with large B-cell lymphoma. ...

Improvements in medical care have reduced the rate of relapse in acute myeloid leukemia after transplants, helped people live...
A new study found that a decrease in a specific population of circulating tumor cells over the first two...
The FDA approved a HER2-targeted therapy for the treatment of breast tumors with low HER2 expression. The U.S. Food...
“Patient-centered” study seeks to assess quality of time that patients live by measuring the “treatment-free survival” outcome. A treatment...

Lorazepam used to relieve stress may cause inflammation that promotes tumor growth and shortens progression-free survival, study says. A...

Finding the latest discoveries in breast cancer care together. More than 7,000 breast cancer researchers, survivors, advocates, and others...
The FDA has approved the ALK inhibitor alectinib for surgically resectable NSCLC. The U.S. Food and Drug Administration (FDA)...
The FDA has approved the FGFR inhibitor erdafitinib for certain patients with urothelial carcinoma The U.S. Food and Drug...
The U.S. FDA approved the combination of lenvatinib and pembrolizumab to treat adult patients with renal cell carcinoma, the...
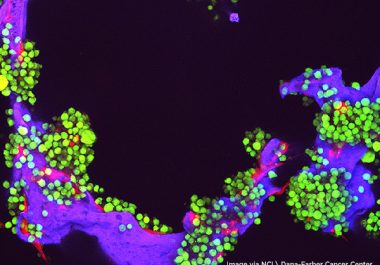
The FDA approved the molecularly targeted therapeutic selinexor to be used in combination with another targeted agent and a...
Enfortumab vedotin-ejfv plus pembrolizumab was approved for certain bladder cancers. The U.S. Food and Drug Administration (FDA) has approved...
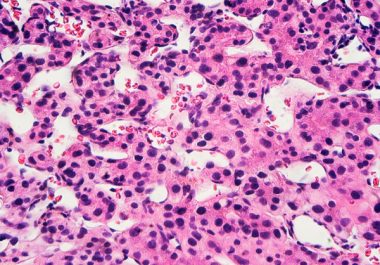
The FDA approved a combination of an immunotherapy and a therapeutic that can stop tumors from growing blood vessels...
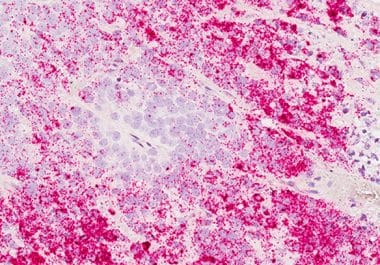
The FDA approved abemaciclib with endocrine therapy for HR-positive, HER2-negative, early-stage breast cancer at high risk of recurrence. The...
The FDA has approved capivasertib for the treatment of breast cancers with certain mutations. The U.S. Food and Drug...
The FDA approved pembrolizumab, in combination with chemotherapy, for certain patients with endometrial cancer. The U.S. Food and Drug...

Clinical outcomes are similar between Black and non-Black patients when patients receive identical treatment and follow-up in a highly...

Lowering dietary fat intake reduced death rates in a subgroup of women with breast cancer who were part of...

A new study showed that one in five young, non-Hispanic white women who tan indoors are dependent on the...
The FDA approved the immune checkpoint inhibitor, cemiplimab-rwlc, for certain patients with locally advanced or metastatic basal cell carcinoma—the...
Belzutifan is now available for certain patients with kidney cancer whether or not they have von Hippel-Lindau disease. The...
A targeted therapy regimen was approved to treat some children with brain cancer The U.S. Food and Drug Administration...

A drug that selectively targets cancer cells was approved for the treatment of certain HR-positive breast cancers. The U.S....
The FDA has approved lutetium Lu 177 dotatate for children 12 years or older with some gastroenteropancreatic neuroendocrine tumors. ...
THE FDA GRANTED ACCELERATED APPROVAL TO A TARGETED THERAPY FOR PATIENTS WITH CML RESISTANT TO OTHER THERAPEUTICS The U.S....
Cancer immunotherapy is making headlines and generating excitement among biomedical researchers, clinicians, patients and their families, and research advocates....
Mirvetuximab soravtansine-gynx was approved for certain ovarian, fallopian tube, and peritoneal cancers. The U.S. Food and Drug Administration (FDA)...