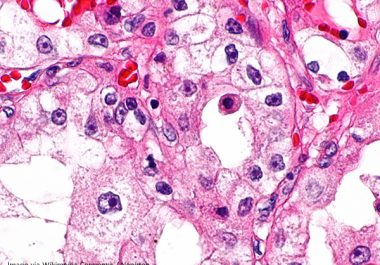
Combining Therapeutics to Treat Kidney Cancer
The FDA approved the combination of an immune checkpoint inhibitor and a therapeutic that prevents tumors from growing blood...

The FDA approved the combination of an immune checkpoint inhibitor and a therapeutic that prevents tumors from growing blood...
The FDA has approved a new targeted therapeutic for certain patients with HER2-positive breast cancer who were previously treated with other targeted therapies....
The U.S. FDA approved a monoclonal antibody and enzyme combination for use in conjunction with chemotherapy and a steroid...
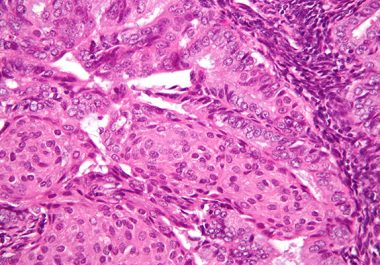
The U.S. FDA approved a combination of a molecularly targeted therapeutic and an immunotherapeutic to treat certain patients with...
The FDA has approved osimertinib plus chemotherapy for non-small cell lung cancer with EGFR mutations. The U.S. Food and...
The FDA has approved amivantamab-vmjw, both alone and in combination with chemotherapy, for the treatment of certain non-small cell...
The FDA approved the use of an immune checkpoint inhibitor as a first-line treatment for patients with colorectal cancers...
The FDA approved the molecularly targeted therapeutic tivozanib to treat certain adult patients with the most common type of...

The former president, who announced in August 2015 he had metastatic melanoma, recently said he was cancer-free after treatment...
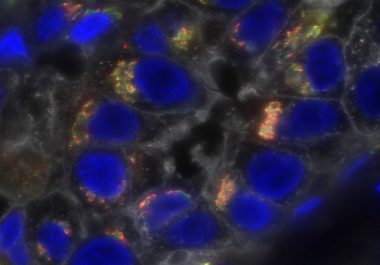
The FDA approved the use of an immunotherapeutic in combination with chemotherapy to treat certain patients with triple-negative breast...
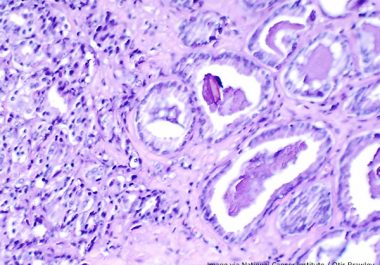
The FDA has expanded the use of the PARP-targeted therapeutic olaparib to include the treatment of certain patients with...

Progression-free survival among certain patients with metastatic colorectal cancer continued at a high level after immune checkpoint inhibitor therapy...

Results from two clinical trials highlight the benefits of the molecularly targeted therapeutics ceritinib and osimertinib There has been...
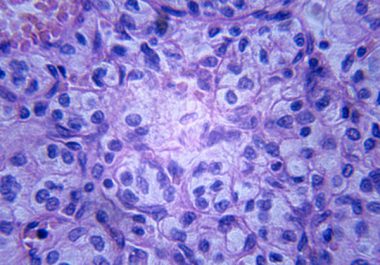
The U.S. Food and Drug Administration approval provides a new option for patients with the most common type of...

The FDA-approved diagnostic test uses blood samples to look for specific mutations in certain patients with metastatic lung cancer....
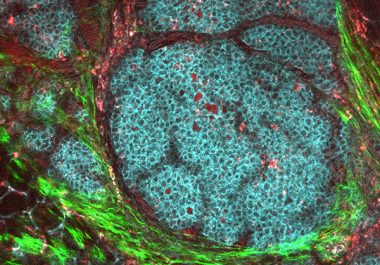
The FDA approved a molecularly targeted therapeutic for postmenopausal women with HR-positive, HER2-negative breast cancer. The U.S. Food and...

The U.S. Food and Drug Administration approval of niraparib means there are now three PARP inhibitors that can be...

Patients with lung cancer who received an immune checkpoint inhibitor before surgery had improved long-term survival rates and lower...
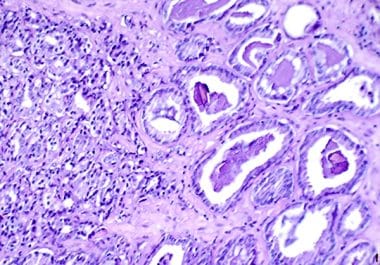
The FDA approved a combination of the PARP inhibitor niraparib with a hormone therapy agent for certain patients with...
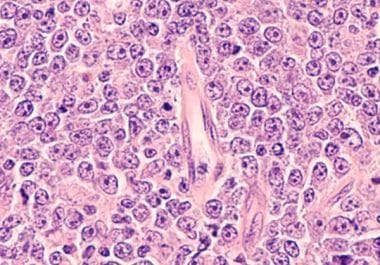
The FDA approved the cellular immunotherapy axicabtagene ciloleucel for second-line treatment of patients with large B-cell lymphoma. The U.S....
The FDA has granted full approval to an antibody-drug conjugate for certain patients with breast cancer. The U.S. Food...

First-line nivolumab with chemotherapy was approved for bladder cancer. The U.S. Food and Drug Administration (FDA) has approved nivolumab...
The FDA has approved a new molecularly targeted therapeutic to treat breast cancer patients with a specific subtype of...
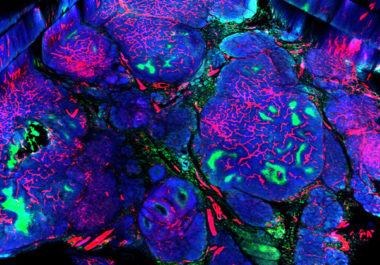
The FDA has expanded the use of the molecularly targeted therapeutic neratinib to include the treatment of certain patients...
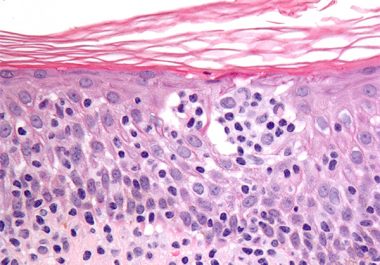
The FDA has expanded the use of a CD30-targeted antibody?drug conjugate to include two additional types of lymphoma. The...
The FDA approval of calaspargase pegol-mknl provides a longer-acting chemotherapy option for certain patients with acute lymphoblastic leukemia. The...

Study presented at the AACR’s eighth annual cancer health disparities conference examines how improved diet reduced the risk of...

The FDA expanded the indications for pembrolizumab to include more patients with stage III-IVA cervical cancer. The U.S. Food...

Fam-trastuzumab deruxtecan-nxki was approved for HER2-positive tumors, regardless of the tissue of origin. The U.S. Food and Drug Administration...
The FDA has approved a new molecularly targeted therapeutic called duvelisib for treating certain patients with blood cancer. The...