A New Targeted Therapy Option for Lung Cancer
The FDA adds a fifth molecularly targeted therapeutic option for the treatment of lung cancer driven by EGFR mutations....

The FDA adds a fifth molecularly targeted therapeutic option for the treatment of lung cancer driven by EGFR mutations....
The amount of residual tumor left after surgery is an important prognostic factor for ovarian cancer patient survival. A...

A recent study reports that how pediatricians talk to parents can affect whether or not they chose to have...
The FDA granted approval to expand the use of a minimally invasive biomarker test to identify certain patients with ovarian,...
A new study suggests that a HER2-targeted therapy approved for breast cancer may be effective for several other cancers....

Clinical trial results suggest patients with high-risk chronic lymphocytic leukemia may benefit from a combination of two targeted therapeutics....
The FDA has approved the immunotherapeutic pembrolizumab to treat Hodgkin lymphoma. The U.S. Food and Drug Administration (FDA) recently...
The FDA’s new approval for the immunotherapeutic pembrolizumab is the first based entirely on a tumor biomarker rather than...
The immune checkpoint inhibitor durvalumab was approved in combination with chemotherapy for certain biliary tract cancers that have spread. ...

Compared to Urbanites, Rural Residents Tend to Think Little Can Be Done about Cancer and Feel Overloaded With Cancer-related...
The FDA has approved repotrectinib for certain patients with non-small cell lung cancer harboring a ROS1 mutation or rearrangement. ...
The U.S. FDA approved a new chemotherapy as part of a multi-drug regimen for certain patients with two similar...
A second inhibitor of the difficult-to-target KRAS protein was approved to treat certain lung cancers. The U.S. Food and...
The FDA has approved the first targeted therapeutic against B-cell maturation antigen (BCMA) to treat certain patients with multiple...

The FDA granted full approval to selpercatinib for the treatment of patients with advanced lung cancer. The U.S. Food...
The U.S. FDA approved an antibody-drug conjugate to treat urothelial cancer, the most common form of bladder cancer. The...
The FDA approved the BRAF inhibitor tovorafenib for pediatric patients with certain Genetic alterations. The U.S. Food and Drug...
The FDA has approved a new immunotherapeutic for treating certain adult patients with diffuse large B-cell lymphoma. The U.S....
The FDA approved a CAR T-cell therapy – a class of cancer immunotherapy – to treat certain patients with...
The FDA has granted accelerated approval to an immunotherapy to treat certain adult patients with endometrial cancer that tests...
The FDA granted accelerated approval to mosunetuzumab-axgb, the first bispecific antibody approved to treat a form of non-Hodgkin lymphoma. ...
The FDA has approved the targeted therapeutic loncastuximab tesirine-lpyl to treat certain adult patients with large B-cell lymphoma. The...
The FDA approved the use of an immune checkpoint inhibitor to treat certain patients with a common form of...
The FDA approved the use of a companion diagnostic to identify patients with tumors that have large numbers of...
The FDA approved the use of an immune checkpoint inhibitor to treat certain patients with cutaneous squamous cell carcinoma. ...

The U.S. FDA approved a molecularly targeted therapeutic for certain patients with tumors associated with a raRe genetic disorder—von...
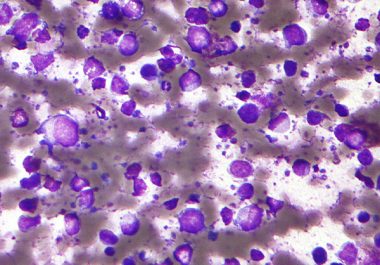
The FDA has approved a targeted therapy drug to treat certain adult patients with diffuse large B-cell lymphoma. The...
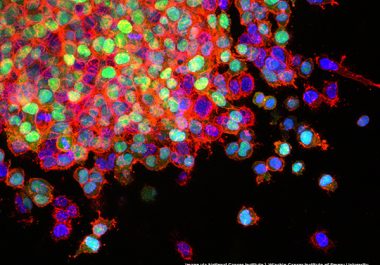
The FDA granted accelerated approval to the first targeted therapeutic and a companion diagnostic for certain adult patients with...
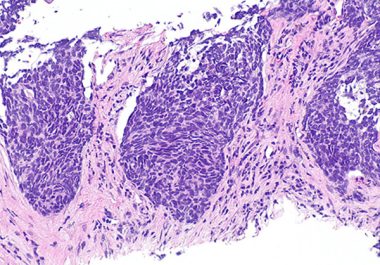
The U.S. FDA has granted accelerated approval to the first KRAS inhibitor, along with two companion diagnostics, for certain...
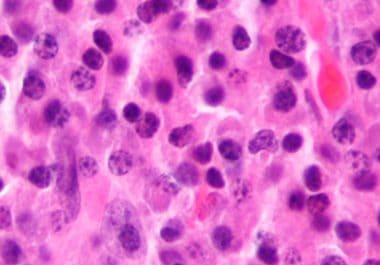
The FDA granted accelerated approval to a targeted immunotherapy for patients with relapsed or refractory multiple myeloma. The U.S....