
Encouraging Muslim Women to Get Mammograms
Study suggests working within religious framework may help ease cultural barriers to cancer screening procedure. Muslim women in America...

Study suggests working within religious framework may help ease cultural barriers to cancer screening procedure. Muslim women in America...
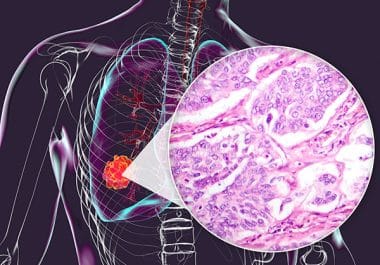
The FDA has approved the ALK inhibitor alectinib for surgically resectable NSCLC. The U.S. Food and Drug Administration (FDA)...

First-line nivolumab with chemotherapy was approved for bladder cancer. The U.S. Food and Drug Administration (FDA) has approved nivolumab...
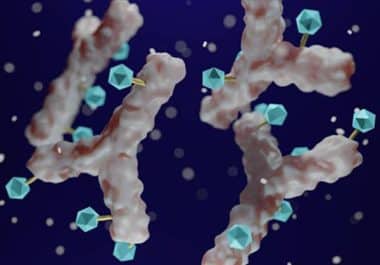
The FDA approved a HER2-targeted therapy for the treatment of breast tumors with low HER2 expression. The U.S. Food...

Trial data first presented at the AACR Annual Meeting 2014, showed new drug doubled progression-free survival for a certain...
“Patient-centered” study seeks to assess quality of time that patients live by measuring the “treatment-free survival” outcome. A treatment...

Lorazepam used to relieve stress may cause inflammation that promotes tumor growth and shortens progression-free survival, study says. A...

The FDA-approved diagnostic test uses blood samples to look for specific mutations in certain patients with metastatic lung cancer....
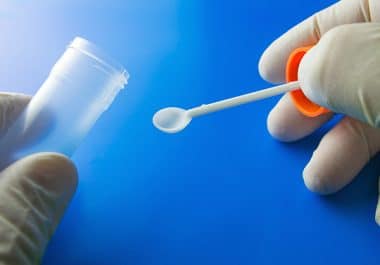
Stool test for signs of cancer, often collected at home, sometimes fails due to inadequate sampling, according to a...
The amount of residual tumor left after surgery is an important prognostic factor for ovarian cancer patient survival. A...
The FDA issued its first approval for the use of A KRAS G12C inhibitor in a tumor type other...
The CAR T-cell therapy received accelerated approval to treat advanced cases of pretreated follicular lymphoma. The U.S. Food and...
Mirvetuximab soravtansine-gynx was approved for certain ovarian, fallopian tube, and peritoneal cancers. The U.S. Food and Drug Administration (FDA)...
The FDA has approved the FGFR inhibitor erdafitinib for certain patients with urothelial carcinoma The U.S. Food and Drug...
The FDA has approved capivasertib for the treatment of breast cancers with certain mutations. The U.S. Food and Drug...

Clinical outcomes are similar between Black and non-Black patients when patients receive identical treatment and follow-up in a highly...

Progression-free survival among certain patients with metastatic colorectal cancer continued at a high level after immune checkpoint inhibitor therapy...

A new genetic biomarker identified breast cancer patients at increased risk for heart damage caused by anthracycline chemotherapy. Women...
The researchers are interested in understanding why some lesions turn into pancreatic cancer to better identify those with a...
The FDA approved the first liquid biopsy companion diagnostic that uses next-generation sequencing to test for EGFR gene mutations...
Tarlatamab-dlle is the first bispecific T-cell engager approved to treat a solid tumor. The U.S. Food and Drug Administration (FDA)...
The FDA approved the RET inhibitor selpercatinib for children with thyroid cancer and other solid tumors harboring various RET...

Tisotumab vedotin-tftv is approved to treat certain recurrent or metastatic cervical cancers. The U.S. Food and Drug Administration (FDA)...
The FDA has approved nogapendekin alfa inbakicept-pmln, which stimulates immune cells to fight bladder cancer. The U.S. Food and...
The RET inhibitor is now fully approved to treat certain patients with RET fusion-positive thyroid cancers. The U.S. Food...
The immunotherapy durvalumab may be used with chemotherapy to treat certain endometrial cancers. The U.S. Food and Drug Administration...
The FDA approved the bispecific T-cell engager blinatumomab for use in the consolidation phase of therapy for certain patients...
The targeted therapy was approved by the FDA for use in combination with bortezomib, lenalidomide, and dexamethasone. The U.S....
The FDA has approved dostarlimab-gxly plus chemotherapy to treat more patients with endometrial cancer. The U.S. Food and Drug...
Tislelizumab-jsgr received its first FDA approval to treat certain esophageal cancers. The U.S. Food and Drug Administration (FDA) has...