A New Option for Breast Cancer Patients
The FDA approved a HER2-targeted therapy for the treatment of breast tumors with low HER2 expression. The U.S. Food...

The FDA approved a HER2-targeted therapy for the treatment of breast tumors with low HER2 expression. The U.S. Food...
“Patient-centered” study seeks to assess quality of time that patients live by measuring the “treatment-free survival” outcome. A treatment...

Lorazepam used to relieve stress may cause inflammation that promotes tumor growth and shortens progression-free survival, study says. A...
The FDA approved a molecularly targeted therapeutic for postmenopausal women with HR-positive, HER2-negative breast cancer. The U.S. Food and...
The FDA approved a combination of the PARP inhibitor niraparib with a hormone therapy agent for certain patients with...
The FDA approved abemaciclib with endocrine therapy for HR-positive, HER2-negative, early-stage breast cancer at high risk of recurrence. The...
The U.S. Food and Drug Administration approval provides a new option for patients with the most common type of...
The FDA has approved capivasertib for the treatment of breast cancers with certain mutations. The U.S. Food and Drug...
The FDA approved pembrolizumab, in combination with chemotherapy, for certain patients with endometrial cancer. The U.S. Food and Drug...

Clinical outcomes are similar between Black and non-Black patients when patients receive identical treatment and follow-up in a highly...

The FDA granted full approval to selpercatinib for the treatment of patients with advanced lung cancer. The U.S. Food...

Screening gaps were largest among racial and ethnic minorities and socioeconomically disadvantaged individuals, study finds. Younger people are less...
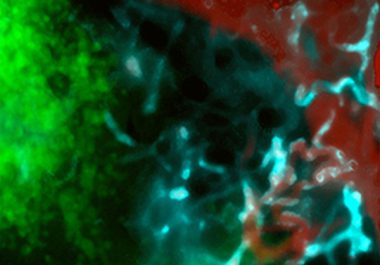
The FDA approval of talazoparib means there are now two PARP inhibitors that can be used to treat patients...
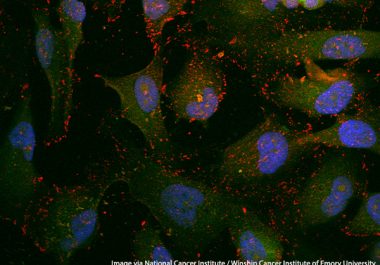
The FDA approved the use of a companion diagnostic to identify patients with tumors that have large numbers of...
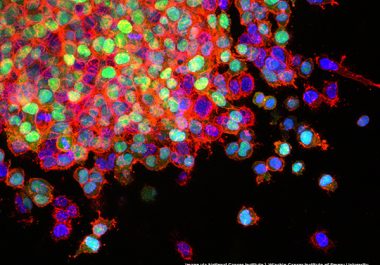
The FDA granted accelerated approval to the first targeted therapeutic and a companion diagnostic for certain adult patients with...
The first FGFR-targeted anticancer therapeutic to be approved by the FDA, erdafitinib, is intended for use in the treatment...
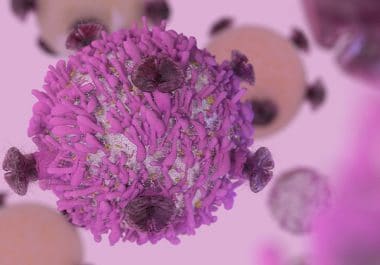
The FDA has approved a new immune checkpoint inhibitor, durvalumab, for the treatment of certain bladder cancer patients. The...

The FDA has approved a combination of nivolumab and ipilimumab to treat certain patients with metastatic lung cancer. The...

“Administrative tasks” for cancer patients are linked to delayed or missed doctor visits, lab tests, prescriptions, and other services. ...
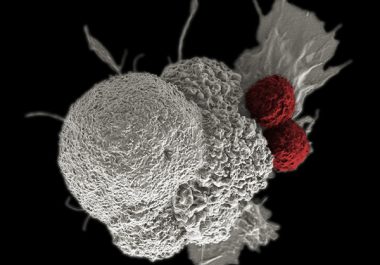
The U.S. Food and Drug Administration approval provides a new treatment option for patients with the most common type...
The FDA recently approved new therapeutics for acute lymphoblastic leukemia, metastatic melanoma, and advanced ovarian cancer. In December 2014,...
The FDA approval makes larotrectinib the first molecularly targeted therapeutic approved for use based on a tumor biomarker rather...

A new genetic biomarker identified breast cancer patients at increased risk for heart damage caused by anthracycline chemotherapy. Women...

Early-onset cancer is increasing globally, testing population-wide preventive DNA testing, and more from the week in cancer news.

Men with metastatic, castrate-resistant prostate cancer with mutations in genes linked to DNA repair responded to targeted therapy. Data...

Drugs and mechanical ventilation appear to be effective in preventing potentially fatal damage to cardiovascular muscles, including those in...
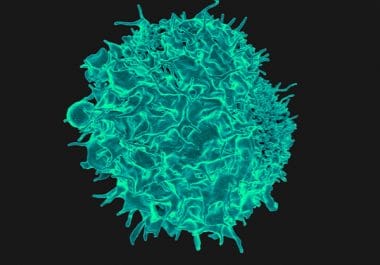
The FDA’s new approval for the immunotherapeutic pembrolizumab is the first based entirely on a tumor biomarker rather than...
The FDA adds a fifth molecularly targeted therapeutic option for the treatment of lung cancer driven by EGFR mutations....

A new study showed that one in five young, non-Hispanic white women who tan indoors are dependent on the...
The FDA has approved the immunotherapeutic pembrolizumab to treat Hodgkin lymphoma. The U.S. Food and Drug Administration (FDA) recently...