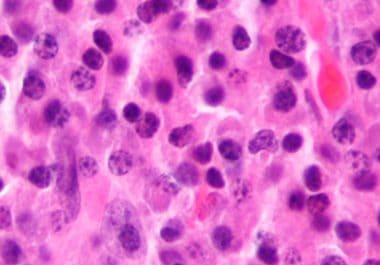
A New CAR T-cell Therapy Option for Multiple Myeloma
The FDA recently approved the second cellular immunotherapy to treat certain adult patients with multiple myeloma. The U.S. Food...

The FDA recently approved the second cellular immunotherapy to treat certain adult patients with multiple myeloma. The U.S. Food...
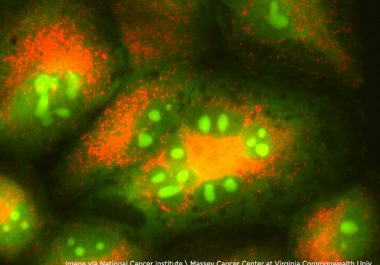
The FDA granted accelerated approval to a new targeted therapeutic and companion diagnostic test for certain adult patients with...
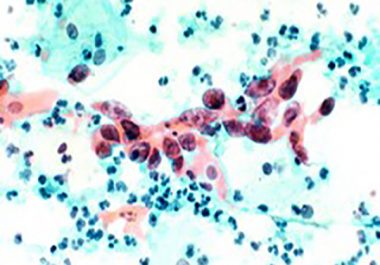
THE FDA GRANTED ACCELERATED APPROVAL TO A NEW THERAPY FOR ADVANCED CERVICAL CANCER The U.S. Food and Drug Administration...
The FDA approved nivolumab in combination with chemotherapy for use before surgery in certain patients with non-small cell lung...
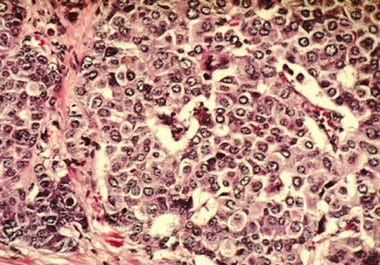
THE FDA APPROVED A NEW TYPE OF CANCER INHIBITOR FOR CERTAIN PATIENTS WITH A HIGH RISK OF RECURRENCE The...
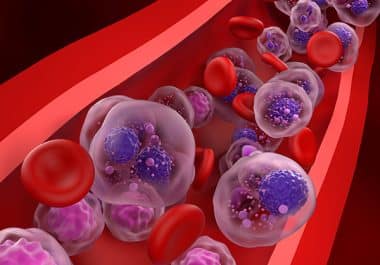
The targeted therapy was approved by the FDA for use in combination with bortezomib, lenalidomide, and dexamethasone. The U.S....
The FDA has approved irinotecan liposome plus chemotherapy for the first-line treatment of metastatic pancreatic cancer. The U.S. Food...
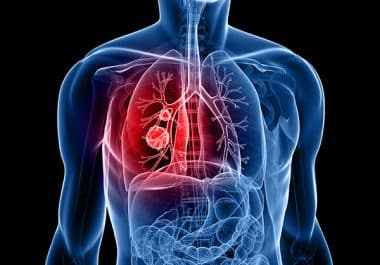
The FDA has granted full approval to tepotinib for some non-small cell lung cancers that harbor a certain mutation...
The FDA approved the combination of two checkpoint inhibitors for certain patients with hepatocellular carcinoma. The U.S. Food and...
The FDA approved a molecularly targeted therapeutic for treating certain patients with some rare cancers affecting the gastrointestinal system. The U.S. Food and...
The FDA has granted accelerated approval to lifileucel, a new type of cell therapy, for the treatment of melanoma....

The FDA granted accelerated approval to a new oral targeted therapeutic to treat certain adult patients with the most...

The FDA approved a combination of trifluridine, tipiracil, and bevacizumab for the treatment of heavily pretreated metastatic colorectal cancer. ...
The FDA granted accelerated approval to a CAR T-cell therapy for certain adult patients with follicular lymphoma. The U.S....
The FDA granted accelerated approval to the molecularly targeted therapeutic umbralisib for certain patients with marginal zone lymphoma or...
The FDA has approved inotuzumab ozogamicin for pediatric patients with certain leukemias. The U.S. Food and Drug Administration (FDA)...
The FDA has approved an immunotherapy to be used in combination with another immunotherapy or chemotherapy for the treatment...
The FDA granted full approval to pralsetinib as a treatment for certain patients with non-small cell lung cancer The...
THE FDA APPROVED THE FIRST ADJUVANT IMMUNOTHERAPY FOR EARLY LUNG CANCER The U.S. Food and Drug Administration (FDA) granted...
Researchers recently developed a risk prediction model that could categorize individuals with lung nodules as having high or low...
The FDA has approved a new molecularly targeted therapeutic for treating certain patients who have advanced gastrointestinal stromal tumors. The...
The FDA has approved a type of molecularly targeted therapeutic called an antibody-drug conjugate for the treatment of metastatic...
The FDA has approved pembrolizumab for the first-line treatment of certain HER2-negative gastric and gastroesophageal junction cancers. The U.S....

Identifying people at the highest risk for pancreatic cancer could better identify those who should be screened to catch...
Approved immunotherapeutics release different brakes on the immune system. A new combination of cancer immunotherapeutics – ipilimumab (Yervoy) and...
The FDA approval is for the use of atezolizumab in combination with Abraxane for the treatment of certain patients...
The FDA has approved a new molecularly targeted therapeutic for treating certain patients with advanced or metastatic HER2-positive breast...
The U.S. Food and Drug Administration approval provides a new option for patients with liposarcoma and leiomyosarcoma that is...
The FDA has approved a combination of the immune checkpoint inhibitor atezolizumab with two targeted therapies for certain patients...

The U.S. Food and Drug Administration approval provides a new option for patients whose disease is driven by mutations...