Targeting a Gene Fusion in Solid Tumors
The FDA granted accelerated approval to selpercatinib to treat solid tumors harboring RET gene fusions. The U.S. Food and...

The FDA granted accelerated approval to selpercatinib to treat solid tumors harboring RET gene fusions. The U.S. Food and...
The FDA has granted accelerated approval to a molecularly targeted therapeutic along with a companion diagnostic to treat certain...
The U.S. FDA granted accelerated approval to an immunotherapeutic and a companion diagnostic test to treat certain cancer patients...
The FDA has approved a new combination of molecularly targeted therapeutics for treating metastatic colorectal cancer with a BRAF...

Multigene panels that rely on next-generation sequencing are increasingly used to test for hereditary cancer risk-related mutations. The federal...
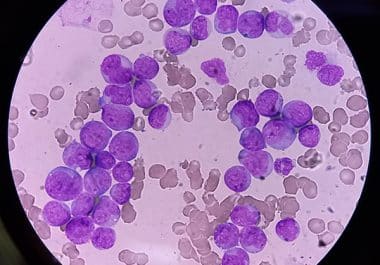
The FDA has approved an inhibitor of FLT3 for use across multiple phases of acute myeloid leukemia treatment. The...

The U.S. Food and Drug Administration approval is intended for patients who have not previously been treated for advanced...

New report charts progress in cancer care, and immediate access to test results can lead to confusion.
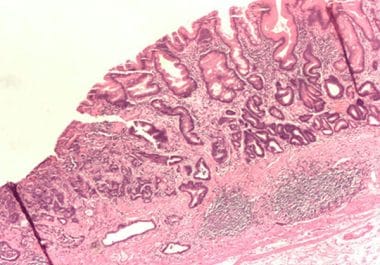
The FDA has approved an immunotherapeutic in combination with chemotherapy to treat certain patients with gastric cancer, gastroesophageal junction...

Prostate cancer is the second most diagnosed cancer among men in the United States, and non-Hispanic black men bear a...
A blood test could help determine who benefits from checkpoint inhibitors in lung cancer, and the FDA approves a...

The FDA granted accelerated approval to a new targeted therapy for certain patients with advanced or metastatic cholangiocarcinoma. The...

The FDA will review a blood test that detects colon cancer, and a writer recounts his sister’s decision to...
THE FDA APPROVED THE IMMUNE CHECKPOINT INHIBITOR PEMBROLIZUMAB IN COMBINATION AND AS A SINGLE AGENT FOR ADVANCED CERVICAL CANCER...

The FDA has approved an immune checkpoint inhibitor in combination with chemotherapy to treat certain adult patients with endometrial...

A new kind of blood test could help detect brain tumors at an earlier stage.
The FDA approved a new regimen that includes a molecularly targeted therapeutic for some older leukemia patients not eligible...

Enasidenib is the second molecularly targeted therapeutic approved by the FDA for treating AML in recent months. The U.S....
The FDA granted full approval to pralsetinib as a treatment for certain patients with non-small cell lung cancer The...
A study found why some patients with mantle cell lymphoma develop resistance to the drug ibrutinib. The finding that...
Two studies in the AACR journal Clinical Cancer Research report that a liquid biopsy test would provide clinicians with...

The FDA has approved a radioactive compound and companion diagnostic test for patients with advanced prostate cancer. The U.S....
The FDA has approved a new HER2-targeted therapeutic for the treatment of certain patients with early-stage breast cancer. The...
The molecularly targeted therapeutic entrectinib is the second therapeutic of its kind approved by the FDA for use based...
The FDA has approved pembrolizumab for treating certain patients with advanced stomach (gastric) cancer. Use of the immunotherapeutic pembrolizumab...
The FDA has approved the IDH1 inhibitor ivosidenib for a group of blood cancers harboring an IDH1 mutation. The...

Researchers are seeking to understand why some patients do not respond to immunotherapeutics and to identify ways to ensure...

People of African descent who receive genetic testing have fewer actionable mutations compared with white people—suggesting fewer available treatment...

Study: Short-term, severe calorie restriction can reduce sugar and other substances in blood that may feed cancer cells and...
Researchers are using genetic tests to help identify breast cancer patients at risk for potentially life-threatening complications of chemotherapy....